Operating under the PRIMO model, CatheGenix offers a product-ready approach, delivering fully FDA-registered solutions that are market-ready while allowing for customization to meet private label and OEM/ODM needs.
We provide professional knowledge in the design, development, and production of high-quality catheters, guide wires, and accessories based on evolving market demands.
With commitment to understanding your unique needs, we tap on our professional experience and large-scale manufacturing capabilities and network to provide complete medical device solutions based on your requirements.
We appreciate any global partners to contact us for further cooperation and build a bright future together.
Over 9000m² of workshops and laboratories, including 4000m² of 10,000-level clean workshop.

Over 10 years’ top and experienced R&D team in design, development and production of medical tubing and interventional medical devices.
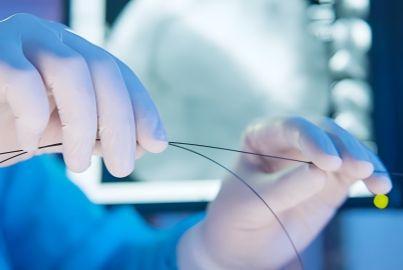
One-stop provider for a variety of catheters, wires & interventional accessories.

The engineering team of hundreds with senior experience and professional background in materials, science, biology, mechanics, chemistry, polymer science and pharmaceutical clinical science.

Millions of capacities with top production capabilities for component manufacturing, breading, precision extrusion, assembly, coating, drug coating and drug formulation.

Industry-leading laboratory level, with a series of high-end laboratory equipment to support the R&D work.

Over 9000m² of workshops and laboratories which certified by ISO13485 etc. Products registration certificates in worldwide (CE, FDA, PMDA).
Equipped with advanced precision extrusion equipment, several certificated testing labs, mass production capacity, excellent ability of component manufacturing and professional R&D team in bio, physics, chemistry, medical field.
We build a bridge for communication and welcome global partners to contact us to discuss PRIMO, CDMO and OEM/ODM cooperations, to foster collaborative innovations and shared business values.